|
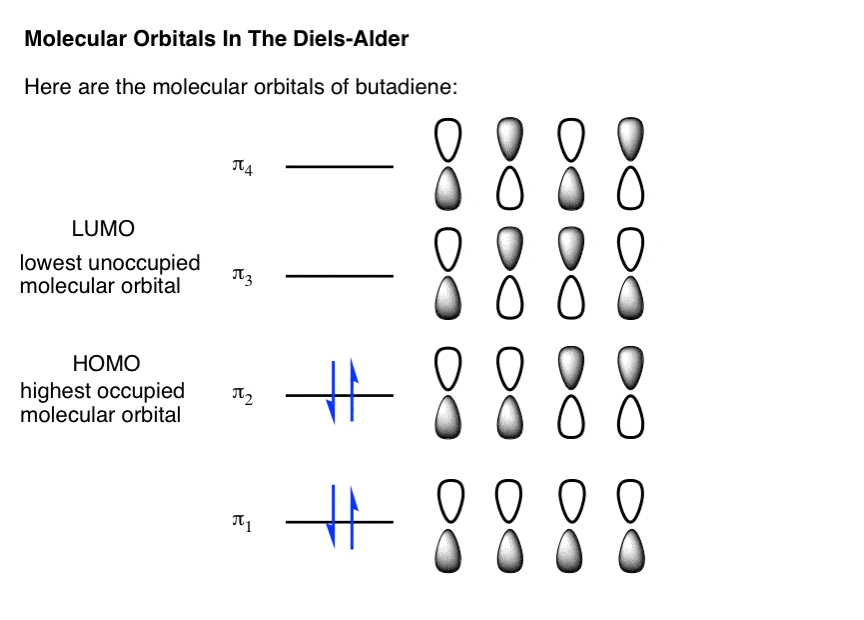
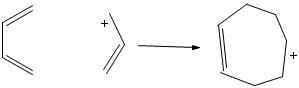
11/10/2022 0 Comments Homo lumo explained Such as chlorophyll, hemoglobin, and cytochrome c. These molecules mimic the active center of biomolecules, Porphyrins and phthalocyanines and their variations have been rather popular as dye molecules for solar cells. Dye-sensitized solar cells are of particular interest here, because they combine a large variety of materials, such as oxide nanoparticles, electrolytes, and organic dye molecules which can be tailored in many different ways. To speed up the development of solar cells with better efficiency at lower cost. This project started with the idea of using sophisticated analysis techniques based on The same reasoning can be appllied to bimolecular pericyclic reactions like the Diels-Alder cycloaddition.New Materials for Solar Cells New Materials for Solar Cells In short, all heterolytic reactions proceed because the energy of a pair of electrons is lowered by the interaction of a filled atomic or molecular orbital with an empty one. You should need no reminder that the carbocation is stabilized by a filled-empty interaction between the empty p orbital of the positive carbon and the s orbital of an adjacent C-H or C-C bond Other examples include the reaction of alkenes with H-X, where the HOMO is the p MO of the alkene and the LUMO is the H-X s* orbital:Īnd the capture of the carobcation in an SN1 reaction by nucleophile: The interaction stabilizes the unshared pair of the oxygen, while simultaneously breaking the CH 3-Cl bond because the interaction is with the antibonding orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
